NARCAN
Structure + Design
Lewis Dot Structure
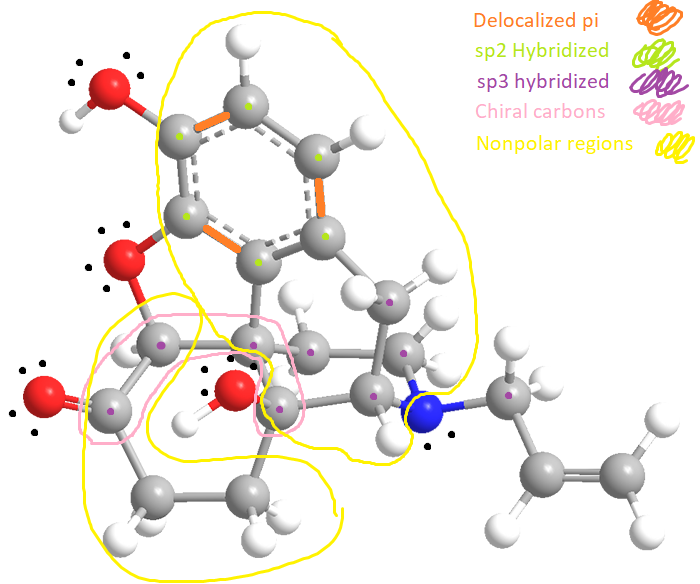
This is the 3-D model Lewis Dot Structure of Narcan
Double bonds are stronger bonds than single bonds because the pull of attraction is stronger due to the bond length being shorter. Typically, the closer two atoms are, the stronger and shorter the bond.The bond length and energy rate includes the following:
-Bond Length (in order from shortest to longest bond length): O-H < C-H < C=C < C-O < C-N < C-C
-Bond Energy (in order from weakest to strongest bond energy): C-N < C-C < C-O < C-H < O-H < C=C
Double bonds are stronger bonds than single bonds because the pull of attraction is stronger due to the bond length being shorter. Typically, the closer two atoms are, the stronger and shorter the bond.The bond length and energy rate includes the following:
-Bond Length (in order from shortest to longest bond length): O-H < C-H < C=C < C-O < C-N < C-C
-Bond Energy (in order from weakest to strongest bond energy): C-N < C-C < C-O < C-H < O-H < C=C
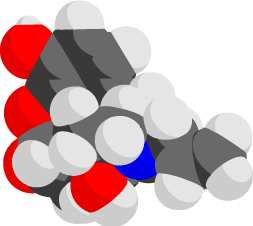
Space-Filling / Electron Density
Here are the following Space Filling model info:
Red = Oxygen (O4)
Blue = Nitrogen (N)
Grey = Carbon (C19)
White = Hydrogen (H21)
Electropositive Regions: Hydrogen (white color)
Electronegative Regions: Oxygen and Nitrogen (Red and Blue colors)
Red = Oxygen (O4)
Blue = Nitrogen (N)
Grey = Carbon (C19)
White = Hydrogen (H21)
Electropositive Regions: Hydrogen (white color)
Electronegative Regions: Oxygen and Nitrogen (Red and Blue colors)